SCENIC+ 的网络研讨会笔记
链接指路:SCENIC+ Stein Aerts lab seminar.
讲解者:Seppe De Winter,文章的二作,来自巨无霸的Stein Aerts Lab - VIB - KULeuven组
安装 Install
这部分真的不想多说,因为众所周知的原因,安装得需要借助强大的“外”力;以及因为“pybigwig==0.3.23”的原因,Windows系统我试了很多次都没有成功。
git clone https://github.com/aertslab/scenicplus
#直播的时候这一步能秒完成,真的馋哭了
#git clone git@github.com:aertslab/scenicplus.git
mamba create --name scenicplus python=3.11.8
#推荐3.11版本
mamba activate scenicplus
cd scenicplus/
git checkout development
pip install .Notes:如果出现下面的报错,原因是git连不上(典)
fatal: unable to access 'https://github.com/aertslab/LoomXpy/': Recv failure: Connection was reset
error: subprocess-exited-with-error
× git clone --filter=blob:none --quiet https://github.com/aertslab/LoomXpy 'C:\Users\Hanoi\AppData\Local\Temp\pip-install-vyuu71m4\loomxpy_152eb1f7ca014b02893fa317b79a7d6c' did not run successfully.
│ exit code: 128
╰─> See above for output.
note: This error originates from a subprocess, and is likely not a problem with pip.
error: subprocess-exited-with-error
× git clone --filter=blob:none --quiet https://github.com/aertslab/LoomXpy 'C:\Users\Hanoi\AppData\Local\Temp\pip-install-vyuu71m4\loomxpy_152eb1f7ca014b02893fa317b79a7d6c' did not run successfully.
│ exit code: 128
╰─> See above for output.
note: This error originates from a subprocess, and is likely not a problem with pip.这个时候我的解决办法是换成美国的VPN(暂时)
有一个十分费力的安装方法,后面得单独开一个页面来讲述了😲
验证安装成功:
终端中直接运行
(base) [chenzhh@nodecw4 ~]$ conda activate scenicplus
(scenicplus) [chenzhh@nodecw4 ~]$ scenicplus
____ ____ _____ _ _ ___ ____
/ ___| / ___| ____| \ | |_ _/ ___| _
\___ \| | | _| | \| || | | _|.|_
___) | |___| |___| |\ || | |__|_..._|
|____/ \____|_____|_| \_|___\____||_|
scenicplus verions: 1.0a2
usage: scenicplus [-h] {init_snakemake,prepare_data,grn_inference} ...
Single-Cell Enhancer-driven gene regulatory Network Inference and Clustering
positional arguments:
{init_snakemake,prepare_data,grn_inference}
options:
-h, --help show this help message and exit
(scenicplus) [chenzhh@nodecw4 ~]$ pycistarget
██████╗ ██╗ ██╗ ██████╗██╗███████╗████████╗ █████╗ ██████╗ ██████╗ ███████╗████████╗
██╔══██╗╚██╗ ██╔╝██╔════╝██║██╔════╝╚══██╔══╝██╔══██╗██╔══██╗██╔════╝ ██╔════╝╚══██╔══╝
██████╔╝ ╚████╔╝ ██║ ██║███████╗ ██║ ███████║██████╔╝██║ ███╗█████╗ ██║
██╔═══╝ ╚██╔╝ ██║ ██║╚════██║ ██║ ██╔══██║██╔══██╗██║ ██║██╔══╝ ██║
██║ ██║ ╚██████╗██║███████║ ██║ ██║ ██║██║ ██║╚██████╔╝███████╗ ██║
╚═╝ ╚═╝ ╚═════╝╚═╝╚══════╝ ╚═╝ ╚═╝ ╚═╝╚═╝ ╚═╝ ╚═════╝ ╚══════╝ ╚═╝
pycistarget version: 1.1
usage: pycistarget [-h] {cistarget,dem} ...
Motif enrichment analysis.
positional arguments:
{cistarget,dem}
options:
-h, --help show this help message and exit
(scenicplus) [chenzhh@nodecw4 ~]$ pycistopic
usage: pycistopic [-h] {qc,topic_modeling,tss} ...
pycistopic: error: the following arguments are required: command**Notes:**不要问为什么pycistopic的输出是个error,因为De Winter讲的时候就是这样样子()
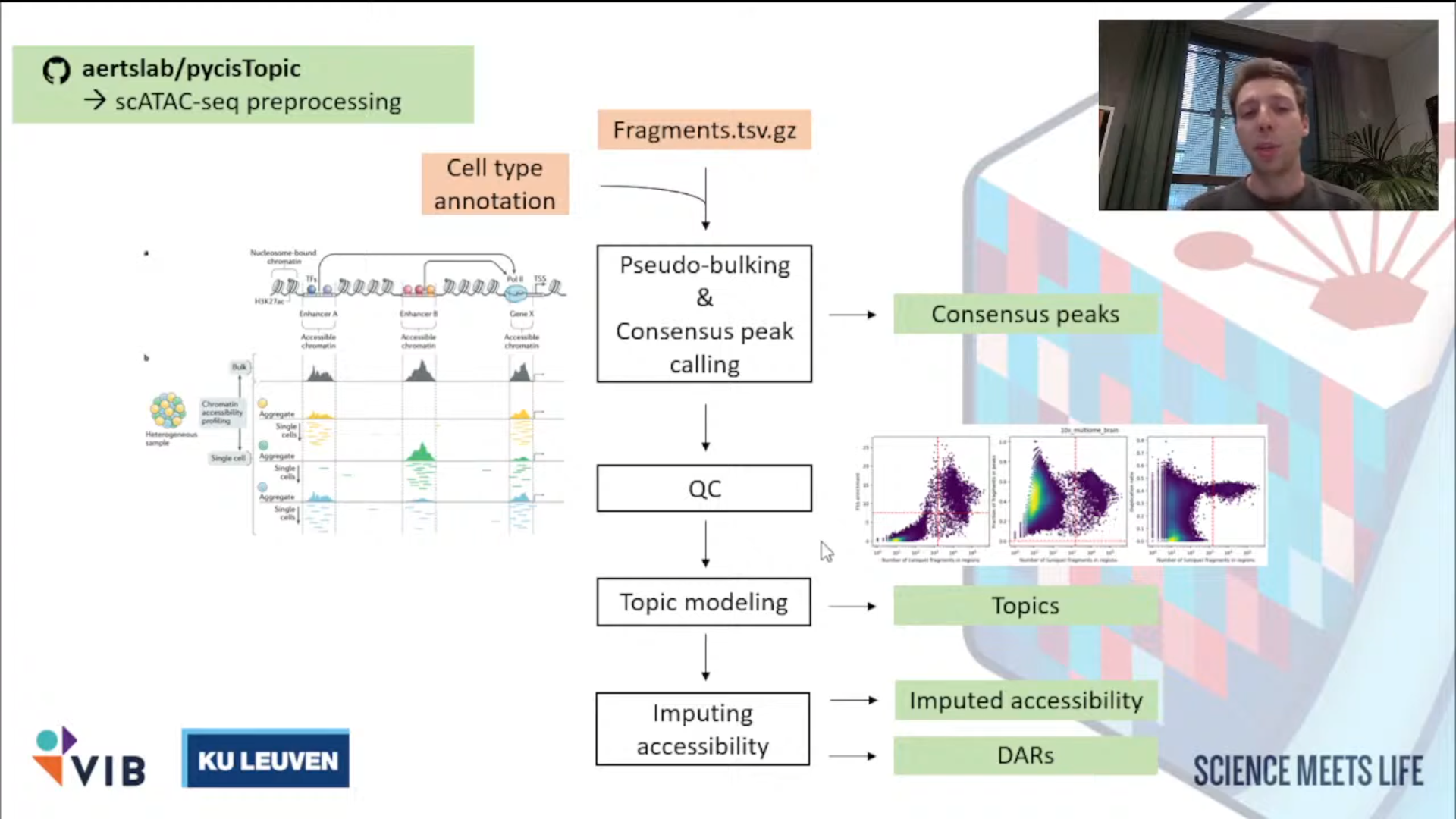
工作流 Workflow Overlook
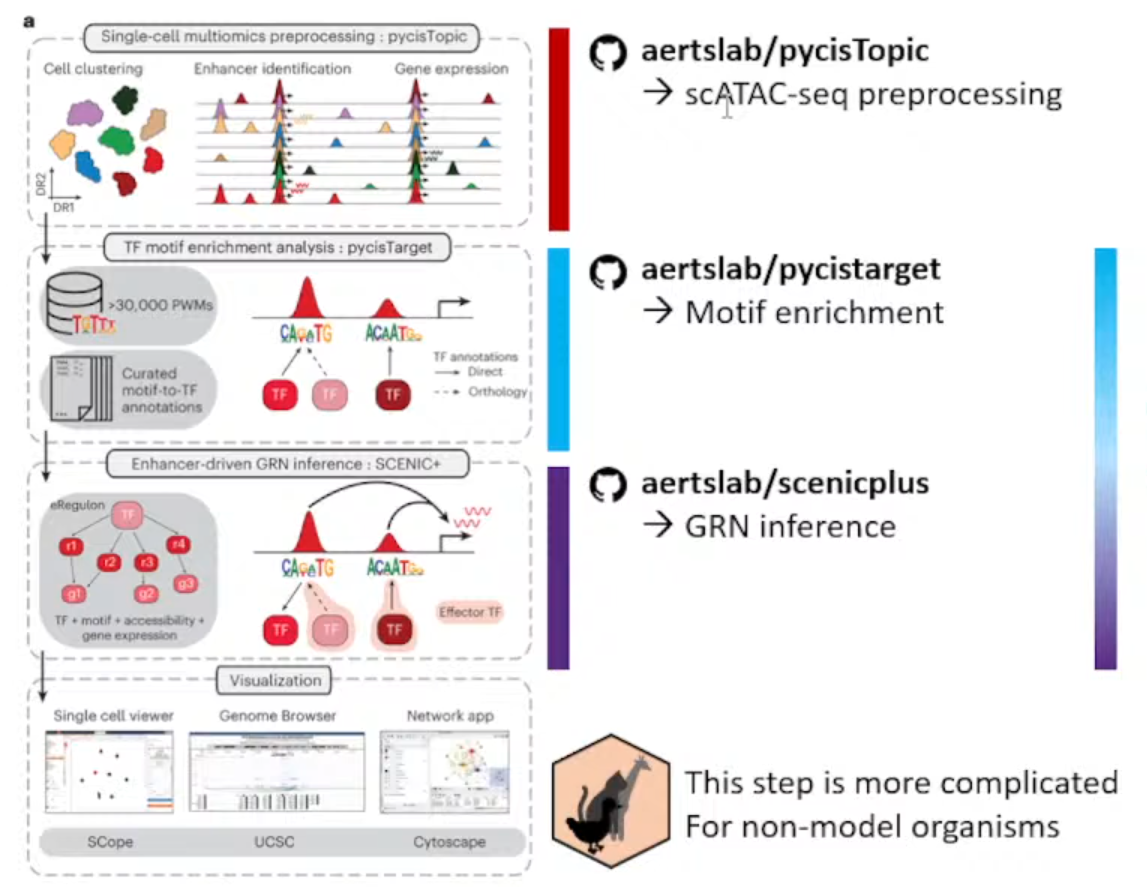
主要目的从ATAC数据+Expression数据(multiomics data)推断enhancer driven GRNs。
主要流程分成了下面三个独立的仓库
①aertslab/pycisTopic
处理ATAC数据,获得consensus peak。
②aertslab/pycisTarget
推断潜在的TF和对应的Region,富集。
③aertslab/scenicplus
结合Expression数据获得GRNs。
Notes:
该方法只推荐用于模式生物和人,一方面是作者只提供了大鼠、小鼠、人和果蝇的blacklist,另一方面可能就是不同生物的GRNs之间真的有很大差异;
pycisTopic
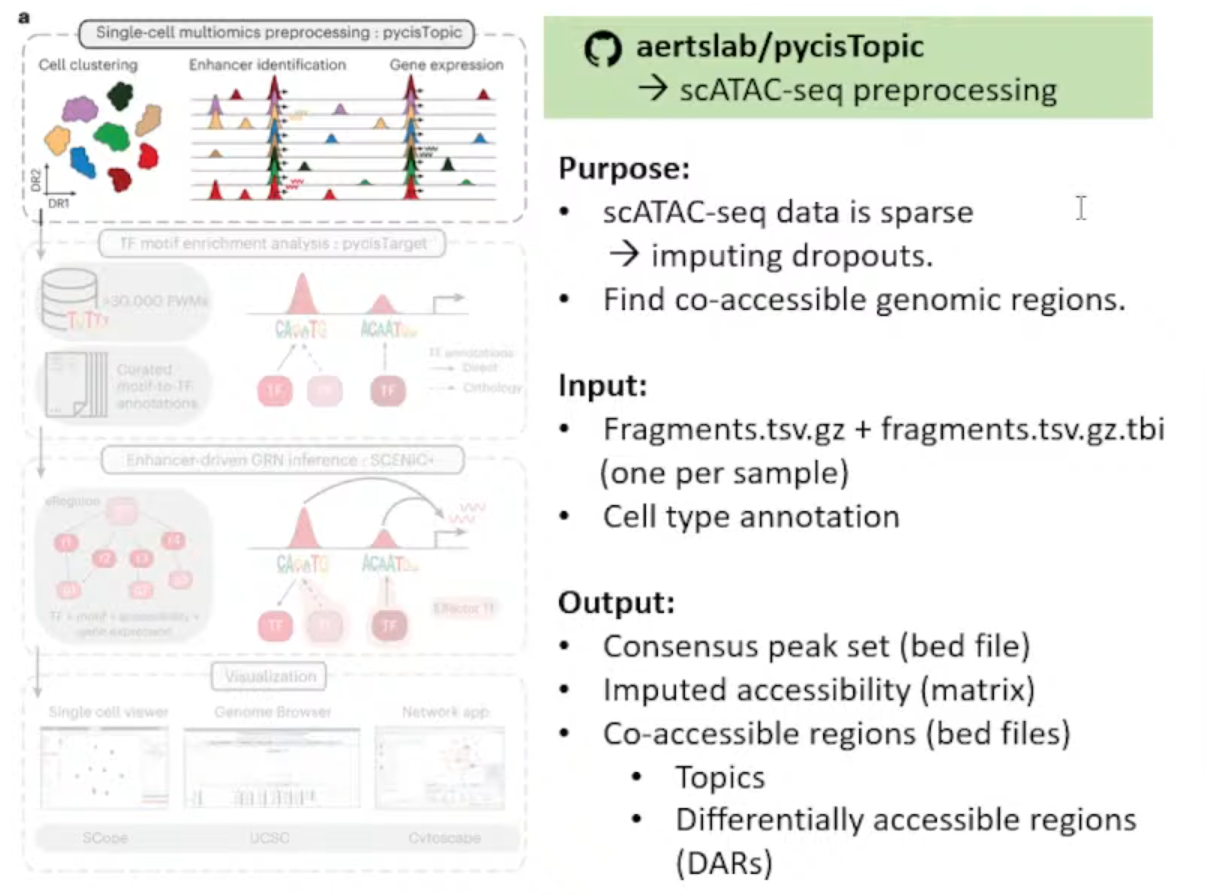
输入文件解释:
①每个样本的Fragmen.tsv.gz文件和index(.tbl)
②细胞注释(Github上有Issue写了无注释的方法,但极不推荐)
生成文件解释:
①bed文件 :存储Consensus peak set
②Imputed accessibility matrix*
Notes:插值的原因是scATAC-seq矩阵高度稀疏(对比scRNA-seq来说)
③bed文件 :存储Co-accessible regions(用于pycisTarget后续分析)
- Topics: 描述染色质的开放模式
- DARs (Differencially accessible regions): 不同细胞类型或条件间有显著差异的区域。
①推断共识峰(获取新的特征)Infer Consensus Peak
<1>Pseudo-Bulk Peak Sets的建立: Aggregate all of the reads coming from Barcodes of the same cell type into a single fragment file.
<2>Call Peaks on each individual fragment file per cell type.
narrow_peak_dict = peak_calling(
macs_path = macs_path,
bed_paths = bed_paths,
outdir = os.path.join(os.path.join(out_dir, "consensus_peak_calling/MACS")),
genome_size = 'hs',
n_cpu = 10,
input_format = 'BEDPE',
shift = 73,
ext_size = 146,
keep_dup = 'all',
q_value = 0.05,
_temp_dir = "/tmp"
)<3>共识整合Consensus peak: Merge all these peaks per cell type into a single Peak Set.
from pycisTopic.iterative_peak_calling import get_consensus_peaks
peak_half_width=250
#peak scale will be set as 500 base
path_to_blacklist="/public/home/chenzhh/packgae_python/pycisTopic/blacklist/hg38-blacklist.v2.bed"
# Get consensus peaks
consensus_peaks = get_consensus_peaks(
narrow_peaks_dict = narrow_peak_dict,
peak_half_width = peak_half_width,
chromsizes = chromsizes,
path_to_blacklist = path_to_blacklist)
#↓导出为bed文件
consensus_peaks.to_bed(
path = os.path.join(out_dir, "consensus_peak_calling/consensus_regions.bed"),
keep =True,
compression = 'infer',
chain = False)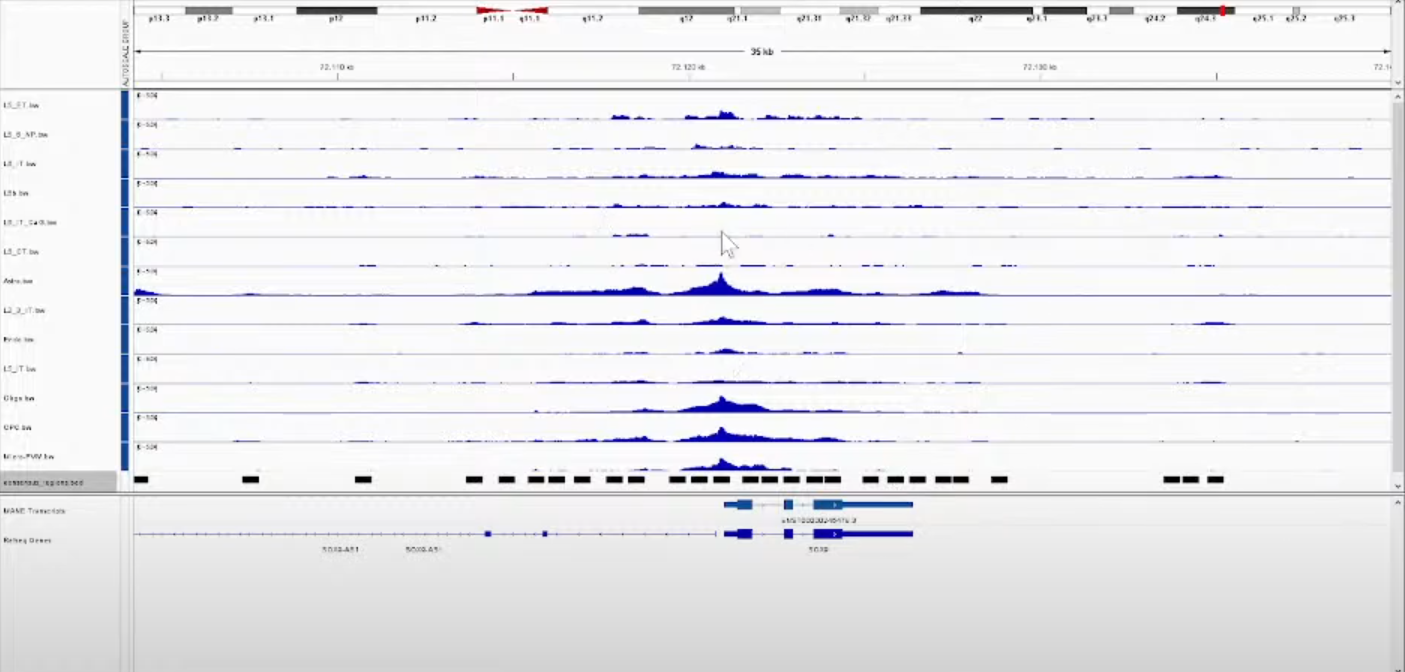
Notes:
使用IGV看得到的结果,其中每一行都是一种细胞类型;
黑色的框则是最后计算得到的共识峰(Consensus peak)。后面的步骤则是以这些Consensus peak作为features,每个Cell Barcodes作为index重新生成matrix。
②QC + 生成新的计数矩阵
filter high quality Cell barcodes(个人觉得,如果之前做过了这步可以省略,而且De Winter讲到这里的时候翻车了hhhh)→generate account matrix
<1>QC
原理和其他工具都一样,简单介绍一下De Winter分享的如何看QC图(本人反正一直都是懵懵懂懂的)
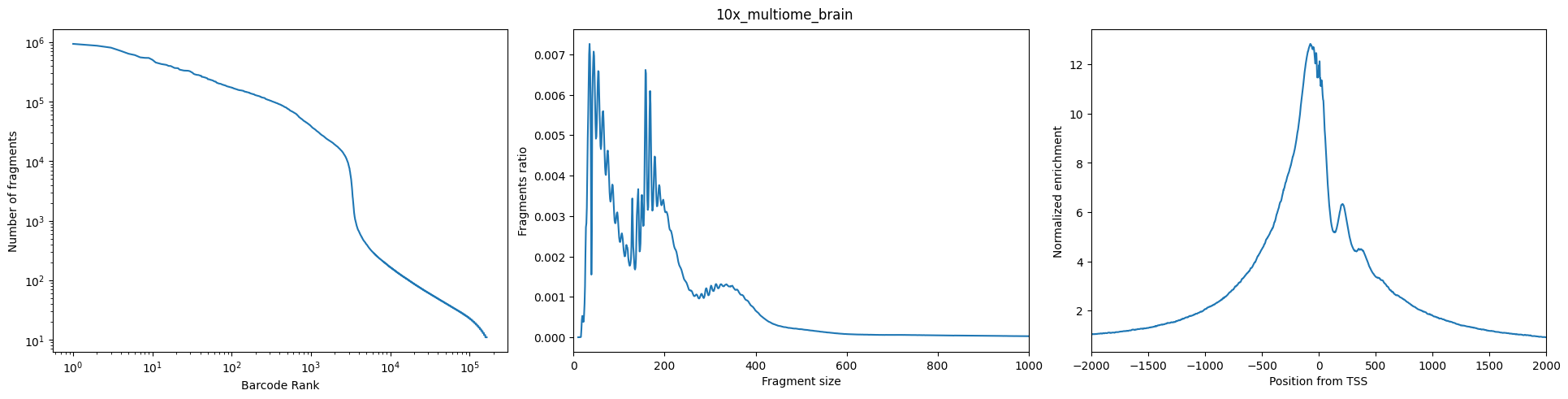
图Left:(Number of Fragment——Barcode Rank): 希望观察到的是一个sharp knee
图Middle(Fragment Ratio——Fragment Size): 长这个样子,两个峰分别代表mononucleosomal (单核小体)和dinucleosomal(双核小体)
图Right(TSS的Normalized Eenrichiment): 很漂亮的TSS enrichment
<2>生成cistopic_obj
pycistopic_qc_output_dir = "outs/qc"
from pycisTopic.cistopic_class import create_cistopic_object_from_fragments
import polars as pl
cistopic_obj_list = []
for sample_id in fragments_dict:
sample_metrics = pl.read_parquet(
os.path.join(pycistopic_qc_output_dir, f'{sample_id}.fragments_stats_per_cb.parquet')
).to_pandas().set_index("CB").loc[ sample_id_to_barcodes_passing_filters[sample_id] ]
cistopic_obj = create_cistopic_object_from_fragments(
path_to_fragments = fragments_dict[sample_id],
path_to_regions = path_to_regions,
path_to_blacklist = path_to_blacklist,
metrics = sample_metrics,
valid_bc = sample_id_to_barcodes_passing_filters[sample_id],
n_cpu = 1,
project = sample_id,
split_pattern = '-'
)
cistopic_obj_list.append(cistopic_obj)如果不使用他们的QC,则将metrics和valid_bc设定为None,如下
path_to_regions = os.path.join(out_dir, "consensus_peak_calling/consensus_regions.bed")
path_to_blacklist = "/public/home/chenzhh/packgae_python/pycisTopic/blacklist/hg38-blacklist.v2.bed"
from pycisTopic.cistopic_class import create_cistopic_object_from_fragments
cistopic_obj_list = []
for sample_id in fragments_dict:
cistopic_obj = create_cistopic_object_from_fragments(
path_to_fragments = fragments_dict[sample_id],
path_to_regions = path_to_regions,
path_to_blacklist = path_to_blacklist,
metrics = None,
valid_bc = None,
n_cpu = 10,
project = sample_id,
split_pattern = '-'
)
cistopic_obj_list.append(cistopic_obj)③Topic modeling
<1>Impute accessibility
<2>获得 Co-accessible Topics: Sets of Co-accessible Regions
pycisTarget
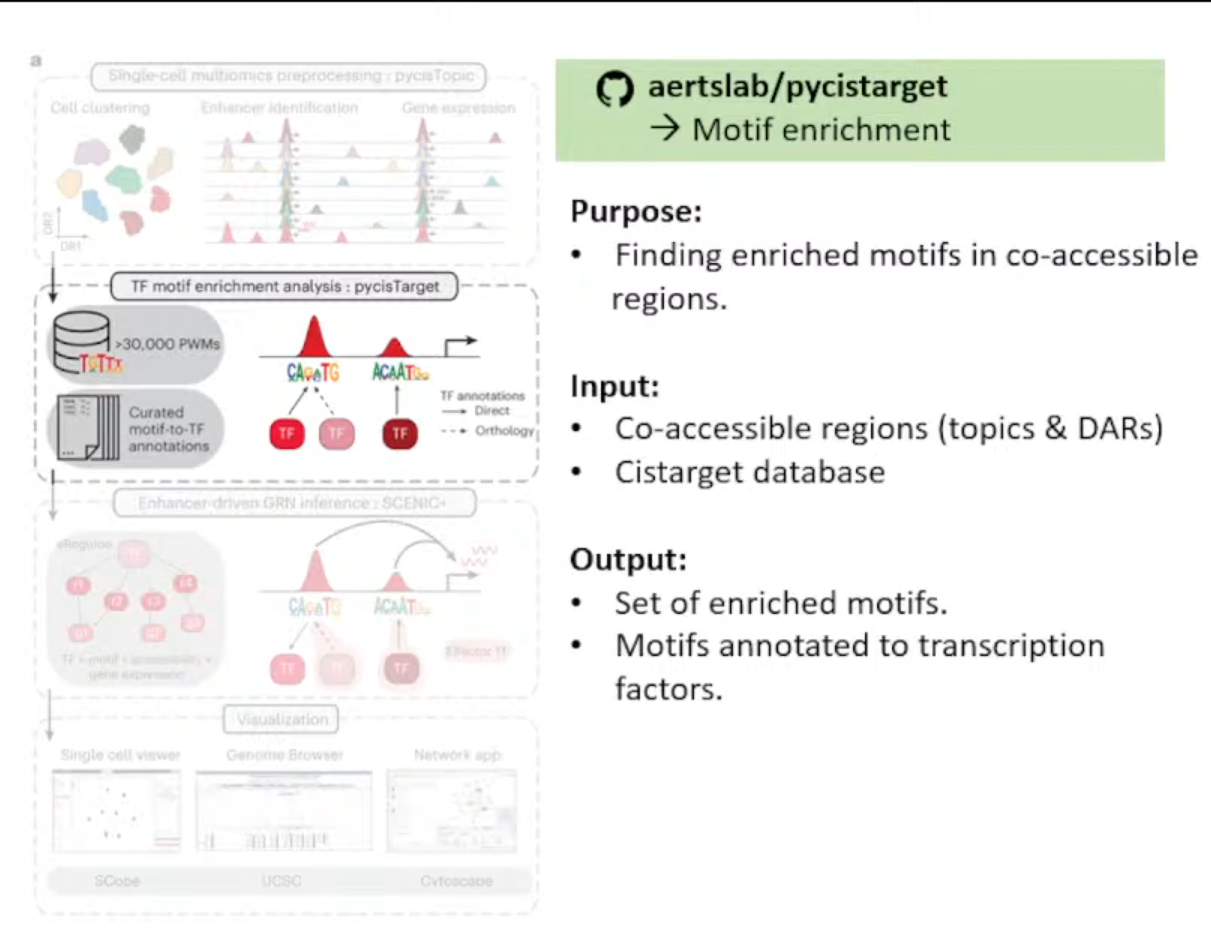
作用:在co-accessible region中找到enrich motifs
输入文件解释:
<1>Set of co-accessible region,可以是Topics或者DARs
→※CisTarget Database
<2>Motif Collection,见网址Index of /cistarget/motif_collections/v10nr_clust_public
输出文件:
注释成TF的富集的motifs集合
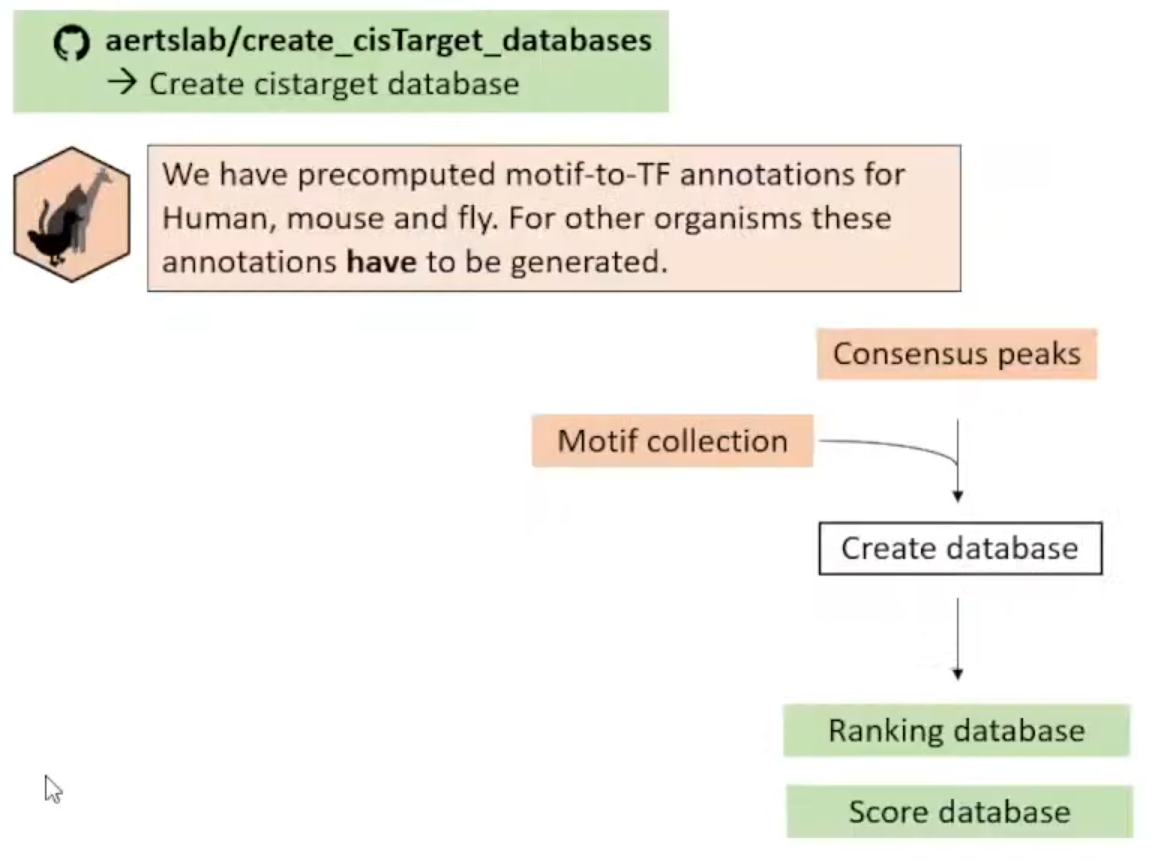
Notes: 尽管他们预先计算好了人鼠果蝇的motifs-to-TF Annotations (Target Database),但这些只是在General regions,所以推荐自己根据Consensus Peaks自己创建Database。
①Create Cistarget Database
<1>访问仓库aertslab/create_cisTarget_databases
给DNA序列进行motifs打分
<2>下载工具Cluster-Buster
使用隐马尔可夫链方法生成分数
wget https://resources.aertslab.org/cistarget/programs/cbust
chmod a+x cbust<3>下载Motif Collection
**<4>**将Consensus Peaks 转为 fasta文件(使用bedtools)
选作: add 1kb of background padding,作为cluster-buster的Background sequence。
<5>运行
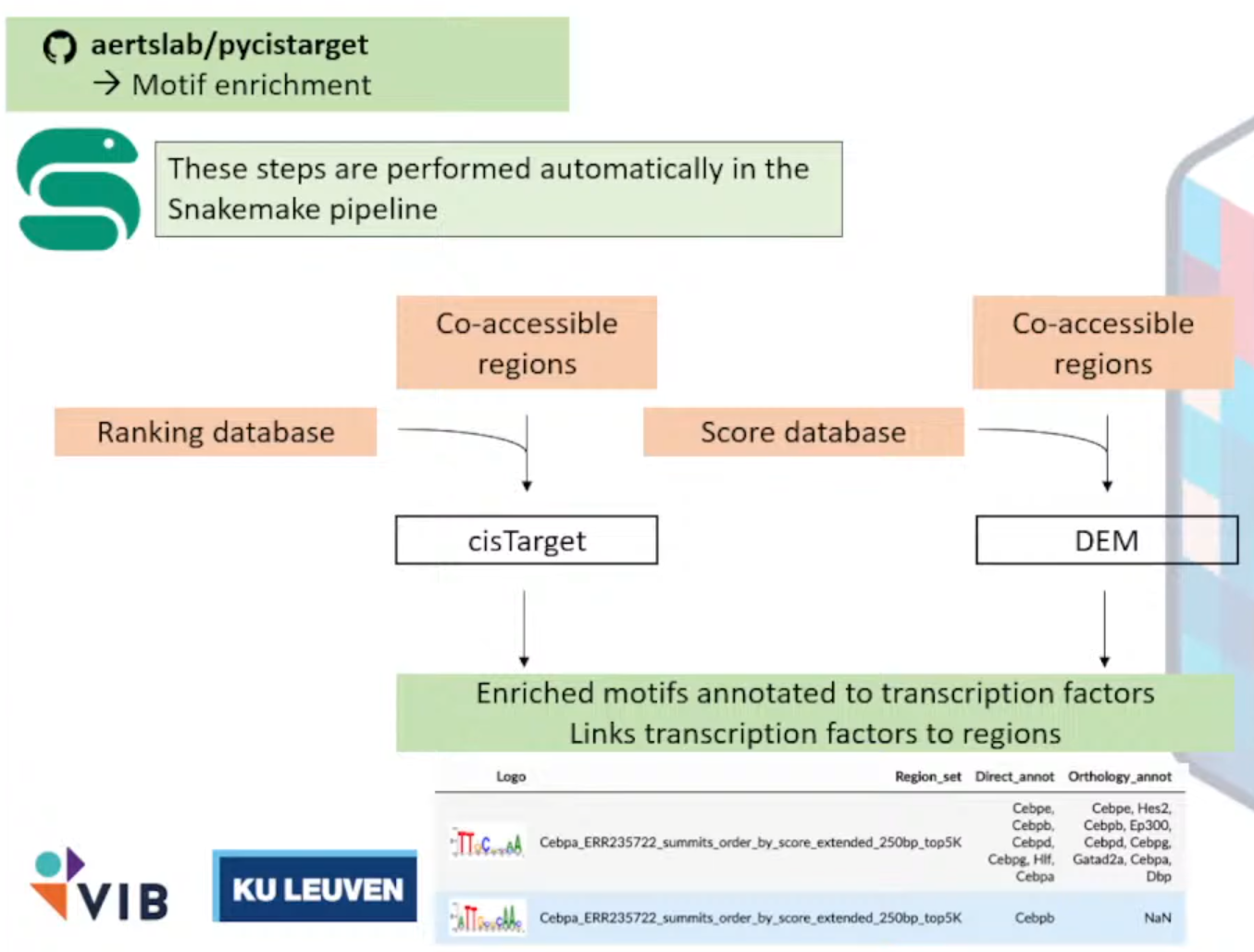
①run_create_fasta.sh
#!/usr/bin/env bash
eval "$(/public/home/chenzhh/bin/micromamba shell hook -s bash)"
micromamba activate create_cistarget_databases
GENOME_FA="/public/home/chenzhh/udanmas/Dianmu/outs/mm10.fa"
CHRSZS="/public/home/chenzhh/udanmas/Dianmu/outs/mm10.chrom.sizes"
REG_BED="/public/home/chenzhh/udanmas/Dianmu/outs/consensus_peak_calling/consensus_regions.bed"
OUT_FA="/public/home/chenzhh/udanmas/Dianmu/outs/mouse_brain_with1kb_bg.fa"
PADDING=1000
SCRIPT_DIR="/public/home/chenzhh/packgae_python/create_cisTarget_databases"
CREATE_SH="${SCRIPT_DIR}/create_fasta_with_padded_bg_from_bed.sh"
command -v bedtools >/dev/null 2>&1 || { echo >&2 "Error: bedtools not found in PATH."; exit 1; }
[ -x "${CREATE_SH}" ] || { echo >&2 "Error: script ${CREATE_SH} not found or not executable."; exit 1; }
echo "[$(date)] Starting create_fasta_with_padded_bg_from_bed..."
bash "${CREATE_SH}" \
"${GENOME_FA}" \
"${CHRSZS}" \
"${REG_BED}" \
"${OUT_FA}" \
"${PADDING}" \
yes
RET=$?
if [ $RET -eq 0 ]; then
echo "[$(date)] Done successfully: ${OUT_FA}"
else
echo "[$(date)] ERROR (exit code $RET)"
fi
exit $RET②
#!/usr/bin/env bash
OUT_DIR="/public/home/chenzhh/udanmas/Dianmu"
SCRIPT_DIR="/public/home/chenzhh/packgae_python/create_cisTarget_databases"
DATABASE_PREFIX="mouse_brain"
FASTA_FILE="${OUT_DIR}/outs/mouse_brain_with1kb_bg.fa"
CBDIR="/public/home/chenzhh/udanmas/Mindulle/aertslab_motif_colleciton/v10nr_clust_public/singletons"
MOTIF_LIST="${OUT_DIR}/motifs.txt"
CORES=180
MICROMAMBA_BIN="/public/home/chenzhh/bin/micromamba"
cd "${OUT_DIR}" || { echo "Cannot cd to ${OUT_DIR}"; exit 1; }
ls "${CBDIR}" > "${MOTIF_LIST}"
if [ ! -x "${SCRIPT_DIR}/create_cistarget_motif_databases.py" ]; then
echo "Error: ${SCRIPT_DIR}/create_cistarget_motif_databases.py not found or not executable"
exit 1
fi
echo "[$(date)] START cisTarget DB build with ${CORES} cores"
"${MICROMAMBA_BIN}" run -n create_cistarget_databases python \
"${SCRIPT_DIR}/create_cistarget_motif_databases.py" \
-f "${FASTA_FILE}" \
-M "${CBDIR}" \
-m "${MOTIF_LIST}" \
-o "${OUT_DIR}/${DATABASE_PREFIX}" \
--bgpadding 1000 \
-t ${CORES}
RET=$?
if [ $RET -eq 0 ]; then
echo "[$(date)] DONE cisTarget DB: ${OUT_DIR}/${DATABASE_PREFIX}"
else
echo "[$(date)] ERROR (exit ${RET})"
fi
exit $RET③运行
chmod +x /public/home/chenzhh/udanmas/Dianmu/run_create_fasta.sh
nohup bash run_create_fasta.sh > create_fasta.log 2>&1 &
tail -f create_fasta.log
chmod +x /public/home/chenzhh/udanmas/Dianmu/run_create_cistarget_db.sh
nohup bash run_create_cistarget_db.sh > create_cistarget_db.log 2>&1 &
tail -f create_cistarget_db.logNotes:
准备环境中需要注意很多很多问题(多到我想紫砂),我觉得后面他们应该会优化,所以我只简单写一下我搞完后还记得的内容吧
#标准创建这个环境请查看github吧,实在没有重复的心情(算了还是记录一遍吧)
git clone -b change_f4_output https://github.com/ghuls/cluster-buster/
cd cluster-buster/
make cbust
#顺带一提,cbust_amd_libm_aocc的编译不是必须的,只是一个加速项,编译这个那可费老鼻子劲了)
cp -a cbust "${CONDA_PREFIX}/bin/cbust"
cd "${CONDA_PREFIX}/bin"
wget http://hgdownload.soe.ucsc.edu/admin/exe/linux.x86_64/liftOver
wget http://hgdownload.soe.ucsc.edu/admin/exe/linux.x86_64/bigWigAverageOverBed
chmod a+x liftOver bigWigAverageOverBed
micromamba activate create_cistarget_databases
mamba install -c bioconda bedtools以及推荐环境安装时,推荐环境代码得到的环境能跑通那就有鬼了,一些很重要的点包括但不限于bedtools应该大于2.31版本他们也是只字未提(默认会直接安装2.14)
下面是我推荐的环境
name: create_cistarget_databases
channels:
- conda-forge
dependencies:
- python=3.10
- numpy=1.21.6
- pandas=1.5.3
- pyarrow>=7.0.0
- numba=0.56.4
- python-flatbuffers
- bedtools=2.31.1SCENICPlus
讲者: Darina Abaffyová, 同组的PhD
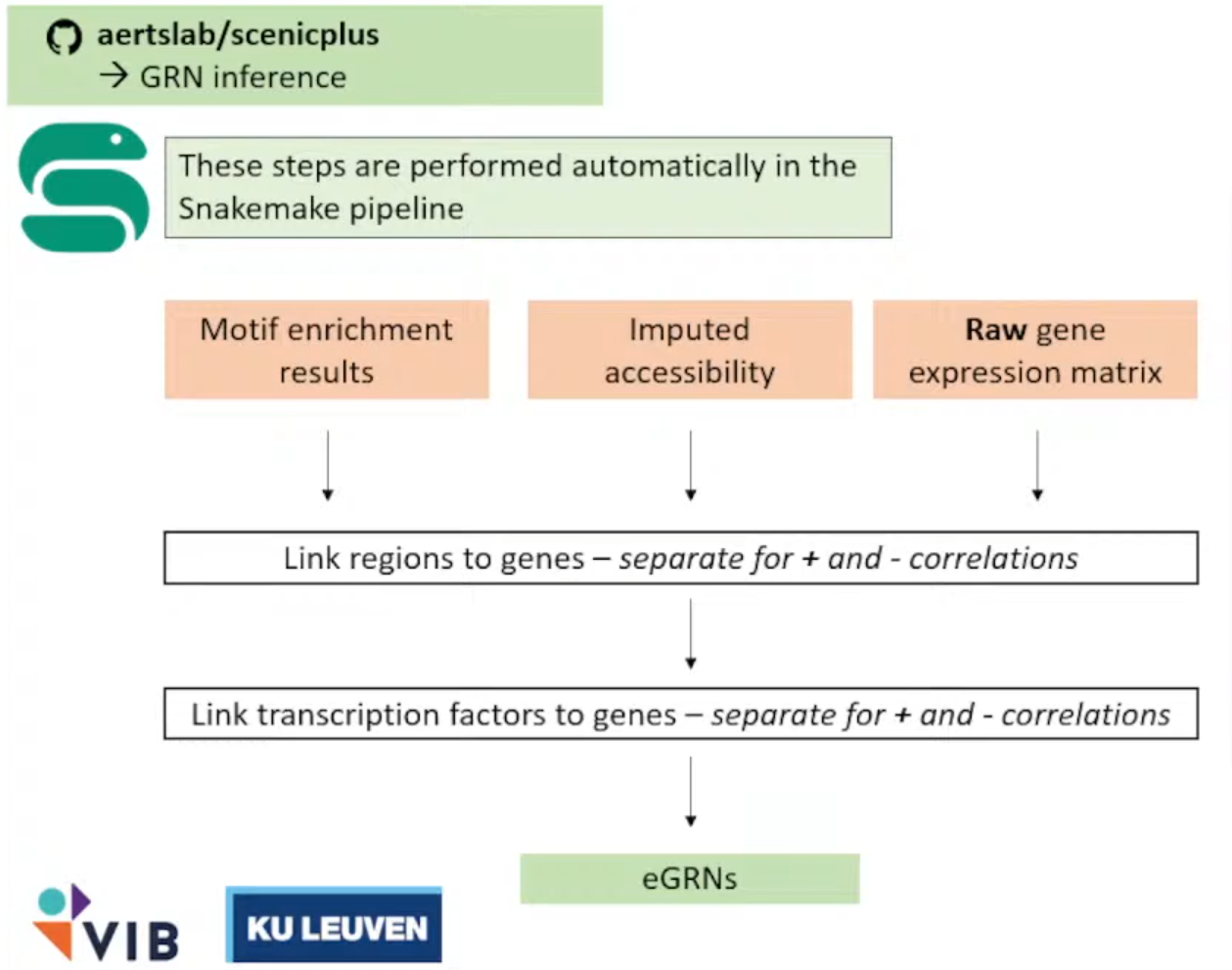
终于啊,终于,我们终于克服万难,但现在scenicplus的流程才刚刚开始(
他们使用了一个Snakemake的工具将整个流程进行封装,这样我们需要做的只有写一个config.ymal文件指向我们的文件(以及无休无止的对输入文件的Debug→因为流程的代码碰也不能碰)
①配置
Snakemake的工作目录是下面这样的:
conda activate scenicplus
cd /public/home/chenzhh/udanmas/Lionrock/formal_scenic
scenicplus init_snakemake --out_dir scplus_pipeline
tree scplus_pipeline
#scplus_pipeline/
#└── Snakemake
# ├── config
# │ └── config.yaml
# └── workflow
# └── Snakefile
mkdir -p outs
mkdir -p tmp
vim scplus_pipeline/Snakemake/config/config.yaml对于这个文件的写入,有以下几个文件需要格外说明:
input_data:
cisTopic_obj_fname: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/outs/cistopic_obj.pkl"
#选定好最佳的lda的model、指定多个topic之后保存的文件。可以暂不进行Normalization、Impute等操作,因为这些都会作为运行的Job;
GEX_anndata_fname: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/rna.h5ad"
#Gene Expression单独拎出来的h5ad文件。barcodes需要与cistopic_obj一致 (cistopic_obj.cell_data.index 和 cistopic_obj.cell_names)
region_set_folder: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/outs/region_sets"
#保存bed文件的目录。这一部分是发生大多报错的十字路口;一方面,需要删除掉所有的空文件`stat`;另一方面文件夹的结构和命名要规范;
ctx_db_fname: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/mm10_screen_v10_clust.regions_vs_motifs.rankings.feather"
#regions_vs_motifs.rankings.feather文件。目前我只跑通了使用官方提供的文件的流程。
dem_db_fname: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/mm10_screen_v10_clust.regions_vs_motifs.scores.feather"
#regions_vs_motifs.rankings.feather文件。目前我只跑通了使用官方提供的文件的流程。
path_to_motif_annotations: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Data/aertslab_motif_colleciton/v10nr_clust_public/snapshots/motifs-v10-nr.mgi-m0.00001-o0.0.tbl"
#motif注释文件,来自官网。
output_data:
# output for prepare_GEX_ACC .h5mu
combined_GEX_ACC_mudata: "ACC_GEX.h5mu"
# output for motif enrichment results .hdf5
dem_result_fname: "dem_results.hdf5"
ctx_result_fname: "ctx_results.hdf5"
# output html for motif enrichment results .html
output_fname_dem_html: "dem_results.html"
output_fname_ctx_html: "ctx_results.html"
# output for prepare_menr .h5ad
cistromes_direct: "cistromes_direct.h5ad"
cistromes_extended: "cistromes_extended.h5ad"
# output tf names .txt
tf_names: "tf_names.txt"
# output for download_genome_annotations .tsv
genome_annotation: "genome_annotation.tsv"
chromsizes: "chromsizes.tsv"
# output for search_space .tsb
search_space: "search_space.tsv"
# output tf_to_gene .tsv
tf_to_gene_adjacencies: "tf_to_gene_adj.tsv"
# output region_to_gene .tsv
region_to_gene_adjacencies: "region_to_gene_adj.tsv"
# output eGRN .tsv
eRegulons_direct: "eRegulon_direct.tsv"
eRegulons_extended: "eRegulons_extended.tsv"
# output AUCell .h5mu
AUCell_direct: "AUCell_direct.h5mu"
AUCell_extended: "AUCell_extended.h5mu"
# output scplus mudata .h5mu
scplus_mdata: "scplusmdata.h5mu"
params_general:
temp_dir: "/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/tmp"
n_cpu: 30
seed: 666
#注意配置这些文件,容易报错。
params_data_preparation:
# Params for prepare_GEX_ACC
bc_transform_func: "\"lambda x: f'{x}'\""
is_multiome: True
key_to_group_by: ""
nr_cells_per_metacells: 10
# Params for prepare_menr
direct_annotation: "Direct_annot"
extended_annotation: "Orthology_annot"
# Params for download_genome_annotations
species: "mmusculus"
#hsapiens或者mmusculus
biomart_host: "http://www.ensembl.org"
#2024年之后统一使用这个网站
# Params for search_space
search_space_upstream: "1000 150000"
search_space_downstream: "1000 150000"
search_space_extend_tss: "10 10"
params_motif_enrichment:
species: "mus_musculus"
#homo_sapiens或者mus_musculus
annotation_version: "v10nr_clust"
motif_similarity_fdr: 0.001
orthologous_identity_threshold: 0.0
annotations_to_use: "Direct_annot Orthology_annot"
fraction_overlap_w_dem_database: 0.4
dem_max_bg_regions: 500
dem_balance_number_of_promoters: True
dem_promoter_space: 1_000
dem_adj_pval_thr: 0.05
dem_log2fc_thr: 1.0
dem_mean_fg_thr: 0.0
dem_motif_hit_thr: 3.0
fraction_overlap_w_ctx_database: 0.4
ctx_auc_threshold: 0.005
ctx_nes_threshold: 3.0
ctx_rank_threshold: 0.05
params_inference:
# Params for tf_to_gene
tf_to_gene_importance_method: "GBM"
# Params regions_to_gene
region_to_gene_importance_method: "GBM"
region_to_gene_correlation_method: "SR"
# Params for eGRN inference
order_regions_to_genes_by: "importance"
order_TFs_to_genes_by: "importance"
gsea_n_perm: 1000
quantile_thresholds_region_to_gene: "0.85 0.90 0.95"
top_n_regionTogenes_per_gene: "5 10 15"
top_n_regionTogenes_per_region: ""
min_regions_per_gene: 0
rho_threshold: 0.05
min_target_genes: 10然后在端口运行:
conda activate scenicplus
snakemake --cores 30 #可以和yaml文件中不一致,经过测试以这个为主或者加入到nohup全家桶→
nohup bash run_create_fasta.sh > create_fasta_$(date +%Y%m%d_%H%M%S)_pandas223-222.log 2>&1 &
nohup bash run_create_cistarget_db.sh > create_db_$(date +%Y%m%d_%H%M%S)_pandas223-222.log 2>&1 &
nohup snakemake --cores 20 --rerun-incomplete > snakemake_$(date +%Y%m%d_%H%M%S).log 2>&1 &②八阿哥
遇到的bug包括但不限于数据不行、库不行;环境不行、人不行……下面我们逐步介绍我们对每个bug的解(妥)决(协);
(1)IndexError: list index out of range
bug一般出现在第一个任务localrule motif_enrichment_cistarget,体现为啥也没说还没开始就出师未捷身先死;
> pwd
/cpfs01/projects-HDD/cfff-afe2df89e32e_HDD/zy_22111220045/Farewell/Lionrock/outs/region_sets/DARs_cell_type
> find . -maxdepth 1 -type f -name "*.bed" -size 0 -print
./ExN-CA2.bed
./ExN-DEGLU-1.bed
#可以得知这两个是空的,直接删除了事。这个是做DAR的结果,这一部分经常容易空。(2)ValueError: Length mismatch: Expected axis has 0 elements, new values have 3 elements
bug表现为:
localrule motif_enrichment_cistarget:
2025-06-04 00:12:07,271 cisTarget INFO Reading cisTarget database
joblib.externals.loky.process_executor._RemoteTraceback:
File "/home/zy_22111220045/miniconda3/envs/scenicplus/lib/python3.11/site-packages/joblib/parallel.py", line 754, in _return_or_raise
raise self._result
ValueError: Length mismatch: Expected axis has 0 elements, new values have 3 elements通过反复测试,问题显然是上一步建库操作的问题。
③结果解释&下游分析
<1>scplus_mdata.uns["direct_e_regulon_metadata"]
| Region | Gene | importance_R2G | rho_R2G | importance_x_rho | importance_x_abs_rho | TF | is_extended | eRegulon_name | Gene_signature_name | Region_signature_name | importance_TF2G | regulation | rho_TF2G | triplet_rank | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | chr5:125638655-125639155 | Tmem132b | 0.000466 | 0.057858 | 0.000027 | 0.000027 | Ahctf1 | False | Ahctf1_direct_+/+ | Ahctf1_direct_+/+_(39g) | Ahctf1_direct_+/+_(42r) | 0.581941 | 1 | 0.106458 | 53472 |
| 1 | chr11:35798818-35799318 | Tenm2 | 0.000077 | 0.416579 | 0.000032 | 0.000032 | Ahctf1 | False | Ahctf1_direct_+/+ | Ahctf1_direct_+/+_(39g) | Ahctf1_direct_+/+_(42r) | 0.614399 | 1 | 0.155765 | 56352 |
| 2 | chr15:8169326-8169826 | Cplane1 | 0.148071 | 0.453076 | 0.067087 | 0.067087 | Ahctf1 | False | Ahctf1_direct_+/+ | Ahctf1_direct_+/+_(39g) | Ahctf1_direct_+/+_(42r) | 0.332098 | 1 | 0.115916 | 5396 |
| 3 | chr16:95702923-95703423 | Brwd1 | 0.064941 | 0.166287 | 0.010799 | 0.010799 | Ahctf1 | False | Ahctf1_direct_+/+ | Ahctf1_direct_+/+_(39g) | Ahctf1_direct_+/+_(42r) | 0.579776 | 1 | 0.073294 | 11180 |
| 4 | chr5:150235924-150236424 | Fry | 0.000058 | 0.054841 | 0.000003 | 0.000003 | Ahctf1 | False | Ahctf1_direct_+/+ | Ahctf1_direct_+/+_(39g) | Ahctf1_direct_+/+_(42r) | 0.606757 | 1 | 0.122811 | 56195 |
| ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... | ... |
| 58282 | chr15:54571087-54571587 | Enpp2 | 0.002650 | -0.357639 | -0.000948 | 0.000948 | Zeb1 | False | Zeb1_direct_-/- | Zeb1_direct_-/-_(32g) | Zeb1_direct_-/-_(48r) | 10.831843 | -1 | -0.386763 | 4901 |
| 58283 | chr19:10261259-10261759 | Myrf | 0.000008 | -0.229283 | -0.000002 | 0.000002 | Zeb1 | False | Zeb1_direct_-/- | Zeb1_direct_-/-_(32g) | Zeb1_direct_-/-_(48r) | 2.413886 | -1 | -0.324805 | 50853 |
| 58284 | chr2:127518216-127518716 | Mal | 0.002490 | -0.479707 | -0.001194 | 0.001194 | Zeb1 | False | Zeb1_direct_-/- | Zeb1_direct_-/-_(32g) | Zeb1_direct_-/-_(48r) | 6.385099 | -1 | -0.371317 | 18872 |
| 58285 | chr1:55449142-55449642 | Plcl1 | 0.001321 | -0.161939 | -0.000214 | 0.000214 | Zeb1 | False | Zeb1_direct_-/- | Zeb1_direct_-/-_(32g) | Zeb1_direct_-/-_(48r) | 1.779930 | -1 | -0.215149 | 45186 |
| 58286 | chr6:85187372-85187872 | Exoc6b | 0.003271 | -0.101657 | -0.000333 | 0.000333 | Zeb1 | False | Zeb1_direct_-/- | Zeb1_direct_-/-_(32g) | Zeb1_direct_-/-_(48r) | 2.747754 | -1 | -0.204703 | 34634 |
58287 rows × 15 columns
<2>
每日一个没用小技巧:
ps -eo user,%cpu --no-headers | awk '{cpu[$1]+=$2} END {for (u in cpu) print u, cpu[u]}' | sort -k2 -nr | head查看是哪个人占了最多的核(八嘎

